What's the difference between RO and DI water purification?

Here's the quick answer: RO and DI filters use different physical reactions to clean water. Reverse Osmosis (RO) is used to partially clean-up tap water to make it roughly 90% to 99% pure. Deionization (DI) filters exchange positive hydrogen and negative hydroxyl molecules for positive and negative contaminant molecules in water. DI filtering and other processes are sometimes referred to as "water polishing."
Understanding the difference between reverse osmosis (RO) and deionized (DI) is important when identifying the right water purification unit for your lab. Having access to high quality water is essential for laboratories to carry out their daily processes and workflows. By taking a closer look at different methods of producing both types of water, RO and DI, you can feel confident in your decision regarding water purification systems.
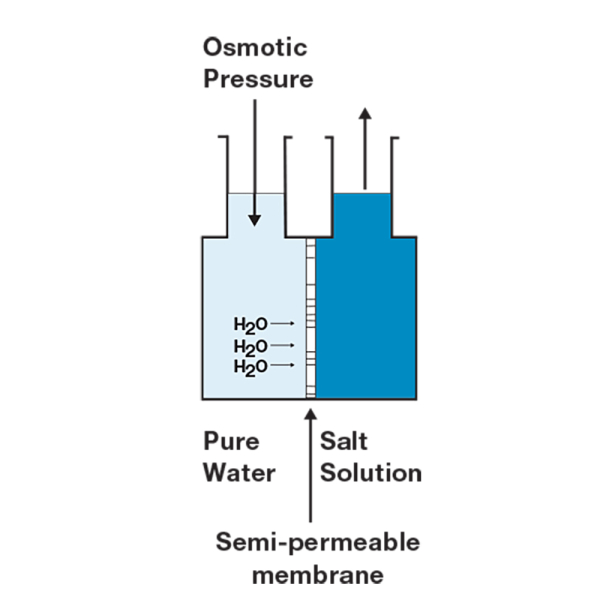
Reverse Osmosis Water
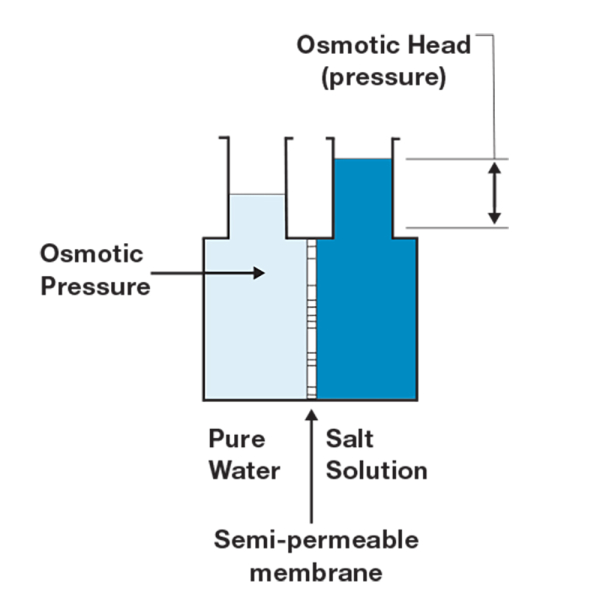
RO grade water, or Type III water, is quite simply the reverse of the naturally occurring process of osmosis. Osmosis is the movement of water molecules from low ion concentration to high ion concentration through a semipermeable membrane. For example, the osmotic process is used by our cells to maintain osmotic balance of the intercellular environment(s). The method of producing RO water is a simple matter of applying increased pressure to one side of the system. By applying additional pressure to one side, in the case the untreated or dirtier water side, feedwater is forced through semipermeable membranes resulting in more purified water. The RO process can typically remove 90-99% of contaminants. Although not perfect, RO purification is a cost-effective method because if used properly RO membranes can last for years.
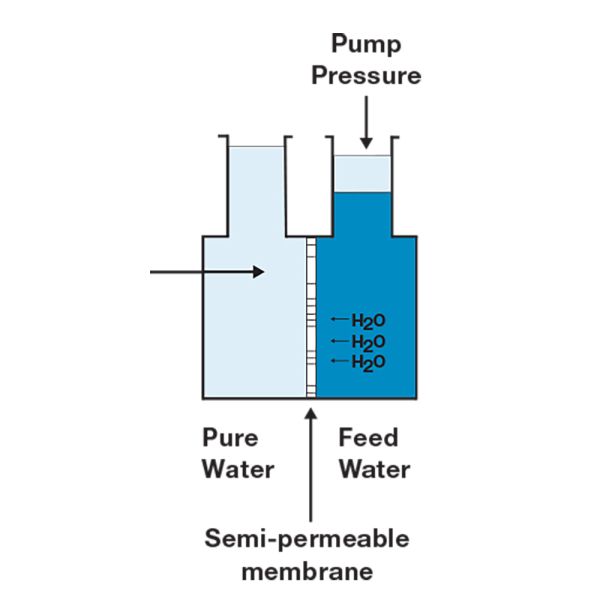
Deionized Water
DI grade water, or Type II water, is purified water that has had almost all its mineral ions removed, such as cations like sodium, calcium, iron, and copper, and anions like chloride and sulfate. The DI process leverages specially manufactured ion-exchange resins that exchange hydrogen (H+) and hydroxyl (OH-) ions for dissolved minerals, and then recombine to form water (H2O). Over time, positively and negatively charged contaminants in the water displace all the active H+ and OH- molecules on the DI resin and at that time the filter must be replaced. Another benefit of the DI purification process is that it is an on-demand process for supplying DI or Type II when needed. Because the ion-exchange resin in not a physical filter with a pore size bacteria and dissolved organics will not be captures, so knowing production water requirements is essential when selecting a water purification unit.
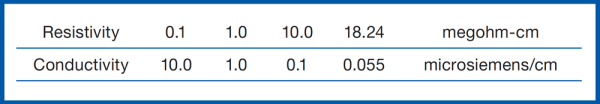
There are several ways to establish the quality level of purified water. The simplest test is a direct measurement of electrical conductivity or resistivity. Most dissolved inorganics are either positively or negatively charged and can transmit an electrical current when electrodes are inserted in the water. The more ions that are present, the greater the conductivity, or the lower the resistivity of the water sample. Conductivity is expressed in µS/cm (microsiemens/cm) and is used to measure water with many ions present.
For example, ocean salt water is packed with dissolved organics and inorganics which provides many opportunities for an electrical current to pass through which corresponds to its 56,000 µS/cm measurement. This isn’t the case for ultrapure Type I water which has ppm level of dissolved organics or inorganics that results in a conductivity or resistivity measurement of 0.055 µS/cm or 18.2 mΩ/cm respectively.
| chevron_left | What type of ductwork do I need? | Articles | The problem with fentanyl: Handling toxic drugs | chevron_right |






