9 things to know before installing an aspiration system
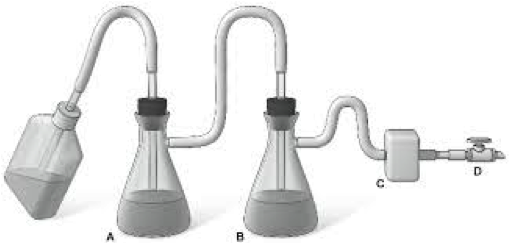 Following “Best-Practices," an Aspiration System for a cell culture lab would look like this: (A) A primary flask with retaining ring or support to prevent tipping, (B) a secondary flask for overflow protection, (D) a vacuum source and a (C) filter between the secondary flask and the vacuum (CDC).
Following “Best-Practices," an Aspiration System for a cell culture lab would look like this: (A) A primary flask with retaining ring or support to prevent tipping, (B) a secondary flask for overflow protection, (D) a vacuum source and a (C) filter between the secondary flask and the vacuum (CDC).
However, these guidelines are frequently overlooked in improvised system set-ups. Often times, the secondary flask is overlooked, the flask(s) are placed on the ground without being anchored, and the filter – if included at all – is rarely replaced or sterilized. When best practices are ignored, a biological risk is posed to the laboratory, its central vacuum system and those working throughout the facility.
Here is what you need to know when investigating an Aspiration System for your laboratory.
PROTECT THE USER – First and foremost an Aspiration System, improvised or purchased, must be designed to protect the operators and technicians in the lab.
1. Systems should include efficient biological filters, such as 0.2µm filters, placed between the receiving flask or bottle and the pump/vacuum system.
2. Receiving bottles should be capable of being autoclaved or sterilized.

PROTECT THE CULTURE – Maintenance of sensitive cultures is difficult, and cleanliness in the work zone is paramount in the successful management of culture lines.
3. All surfaces of the system should be smooth and easy to clean.
4. Tubing should be cleanable or replaceable.
5. If the system will be used by multiple users, then tubing lines should be isolated all the way to the receiving flask.
PROTECT THE LABORATORY – Often times, researchers and technicians have a hyper awareness of their immediate surroundings. Though important, they must not neglect the larger laboratory environment.
6. The receiving flask/bottle should be provided with an appropriate base for support.
7. A primary disinfectant should be used in conjunction with the receiving flask/bottle. If using Bleach as the primary disinfectant in the receiving bottle, ensure that glass is used instead of plastic containers.
MAINTAIN CONTROL – Whether analog, electromechanical or digital, be sure that the level of vacuum you are using matches your needs.
8. When working with sensitive cells or loose pellets, a gentle vacuum should be applied when removing fluids.
9. Large vacuums may be required when removing larger volumes or for higher flow applications.
BVC Fluid Vacuum Aspiration Systems
 The BVC Fluid Aspiration Systems from VACUUBRAND® and Labconco® are convenient to cell culture work and other laboratory application systems. Two types are available: BVC Control Aspiration Systems are for use in labs without in-house vacuum and feature electronic vacuum control. BVC Basic Systems are designed for use with existing vacuum supplies, individual diaphragm pumps and house vacuum systems. Both are available with either Glass or Polypropylene receiving bottles, come with one hand controller and a 0.2µm hydrophobic bio-filter. Contact Labconco for ordering information: 1(800)821-5525 or www.labconco.com.
The BVC Fluid Aspiration Systems from VACUUBRAND® and Labconco® are convenient to cell culture work and other laboratory application systems. Two types are available: BVC Control Aspiration Systems are for use in labs without in-house vacuum and feature electronic vacuum control. BVC Basic Systems are designed for use with existing vacuum supplies, individual diaphragm pumps and house vacuum systems. Both are available with either Glass or Polypropylene receiving bottles, come with one hand controller and a 0.2µm hydrophobic bio-filter. Contact Labconco for ordering information: 1(800)821-5525 or www.labconco.com.
Works Cited
CDC. Biosafety in Microbiological and Biomedical Laboratories. Ed. L. Casey MD Chosewood and Deborah E. Wilson. 5th. Washington, D.C.: National Insitute of Health, 2009.
Fleming, Diane O and Debra L Hunt, Biological Safety: Principles and Practices, 4th Ed. Washington, DC: American Society for Microbiology, 2006.
VACUUBRAND, Inc. "The New Standard in Aspriation Stations." 2013. Document. 12 September 2013.
.
| chevron_left | Video: Easy way to load Stoppering Tray Dryers (Tray with Slide-Out Bottom) | Articles | 3 main steps to fume hood mechanical system selection | chevron_right |






